Ion bonding formula
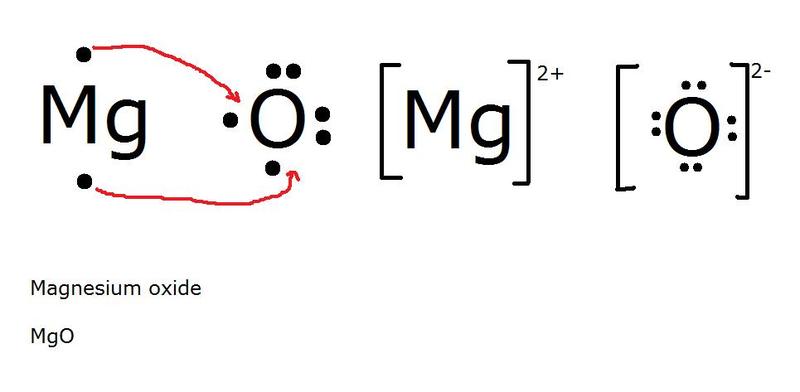
You need one sodium atom to provide the extra electron for one chlorine atom, so they combine together 1:1. The sodium ions and chloride ions are held together by the strong electrostatic attractions between the positive and negative charges. If electrons are gained by an atom, negative ions are formed.Ī negative ion is sometimes called an anion. The chlorine has gained an electron, so it now has one more electron than proton. Positive ions are sometimes called cations. If electrons are lost from an atom, positive ions are formed.
/174898754-56a132333df78cf772684fe7.jpg)
Because it has one more proton than electron, it has a charge of 1+.

The sodium has lost an electron, so it no longer has equal numbers of electrons and protons. If a sodium atom gives an electron to a chlorine atom, both become more stable. If it could gain an electron from somewhere it too would become more stable. If it gave away that electron it would become more stable.Ĭhlorine (2,8,7) has 1 electron short of a stable noble gas structure (2,8,8). Sodium (2,8,1) has 1 electron more than a stable noble gas structure (2,8). We shall have to spend some time later on demolishing the concept! Note: The central role given to noble gas structures is very much an over-simplification.
#ION BONDING FORMULA FULL#
You may well have been left with the strong impression that when other atoms react, they try to organise things such that their outer levels are either completely full or completely empty. These noble gas structures are thought of as being in some way a "desirable" thing for an atom to have. It starts with a simple picture of the formation of ions, and then modifies it slightly for A'level purposes.Īt a simple level (like GCSE) a lot of importance is attached to the electronic structures of noble gases like neon or argon which have eight electrons in their outer energy levels (or two in the case of helium). This page explains what ionic (electrovalent) bonding is.
